L-cysteine: Difference between revisions
(Created page with "== Introduction == L-cysteine is a naturally occurring, sulfur-containing amino acid that plays a critical role in various biological processes. It is classified as a semi-essential amino acid, meaning that while it can be synthesized by the human body, it is also obtained through dietary sources. L-cysteine is involved in protein synthesis, detoxification, and the production of various biomolecules, including glutathione, a powerful antioxidant. == Chemical Struct...") |
No edit summary |
||
| Line 7: | Line 7: | ||
L-cysteine is characterized by the presence of a thiol group (-SH) attached to its side chain, which imparts unique chemical properties. The molecular formula of L-cysteine is C3H7NO2S, and its IUPAC name is (2R)-2-amino-3-sulfanylpropanoic acid. The presence of the thiol group makes L-cysteine highly reactive, allowing it to form disulfide bonds, which are crucial for the structural integrity of proteins. | L-cysteine is characterized by the presence of a thiol group (-SH) attached to its side chain, which imparts unique chemical properties. The molecular formula of L-cysteine is C3H7NO2S, and its IUPAC name is (2R)-2-amino-3-sulfanylpropanoic acid. The presence of the thiol group makes L-cysteine highly reactive, allowing it to form disulfide bonds, which are crucial for the structural integrity of proteins. | ||
[[Image:Detail-97023.jpg|thumb|center|Chemical structure of L-cysteine.|class=only_on_mobile]] | |||
[[Image:Detail-97024.jpg|thumb|center|Chemical structure of L-cysteine.|class=only_on_desktop]] | |||
== Biosynthesis == | == Biosynthesis == | ||
Latest revision as of 09:06, 16 July 2024
Introduction
L-cysteine is a naturally occurring, sulfur-containing amino acid that plays a critical role in various biological processes. It is classified as a semi-essential amino acid, meaning that while it can be synthesized by the human body, it is also obtained through dietary sources. L-cysteine is involved in protein synthesis, detoxification, and the production of various biomolecules, including glutathione, a powerful antioxidant.
Chemical Structure and Properties
L-cysteine is characterized by the presence of a thiol group (-SH) attached to its side chain, which imparts unique chemical properties. The molecular formula of L-cysteine is C3H7NO2S, and its IUPAC name is (2R)-2-amino-3-sulfanylpropanoic acid. The presence of the thiol group makes L-cysteine highly reactive, allowing it to form disulfide bonds, which are crucial for the structural integrity of proteins.
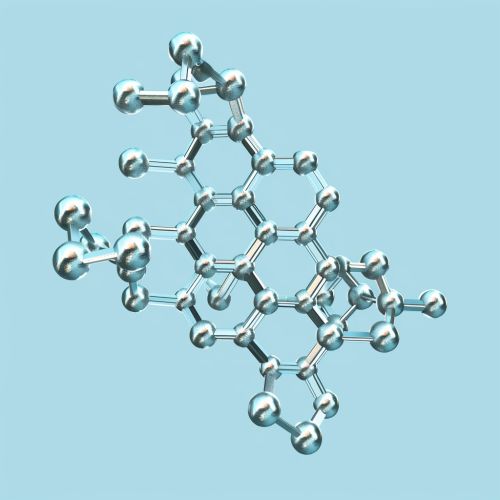
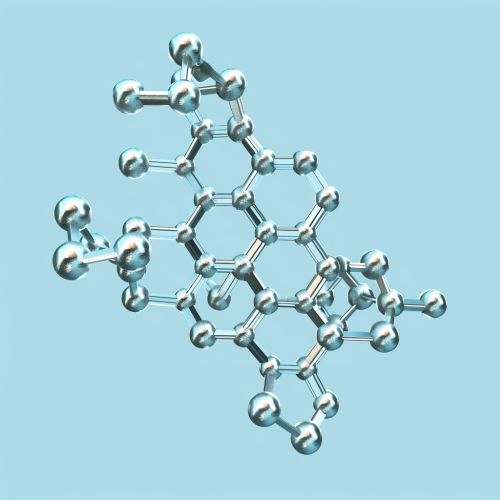
Biosynthesis
L-cysteine is synthesized in the body through the transsulfuration pathway. This pathway involves the conversion of methionine to homocysteine, which is then converted to cystathionine by the enzyme cystathionine beta-synthase. Cystathionine is subsequently cleaved by cystathionine gamma-lyase to produce L-cysteine. The availability of methionine and the activity of these enzymes are critical factors in the regulation of L-cysteine levels in the body.
Biological Functions
Protein Synthesis
L-cysteine is incorporated into proteins during translation, contributing to the formation of disulfide bonds. These bonds are essential for the proper folding and stability of many proteins, particularly those that are secreted or located in the extracellular matrix.
Antioxidant Activity
One of the most significant roles of L-cysteine is its contribution to the synthesis of glutathione, a tripeptide composed of glutamate, cysteine, and glycine. Glutathione acts as a major antioxidant, protecting cells from oxidative stress by neutralizing reactive oxygen species (ROS).
Detoxification
L-cysteine is involved in the detoxification of harmful substances in the liver. It participates in the formation of conjugates with toxins, making them more water-soluble and easier to excrete. This process is facilitated by enzymes such as glutathione S-transferases.
Metabolic Pathways
L-cysteine is a precursor for several important biomolecules, including taurine, coenzyme A, and hydrogen sulfide. Taurine is involved in bile salt formation and osmoregulation, while coenzyme A is essential for fatty acid metabolism. Hydrogen sulfide acts as a signaling molecule with various physiological roles.
Dietary Sources
L-cysteine can be obtained from various dietary sources, including high-protein foods such as meat, poultry, eggs, and dairy products. Plant-based sources include legumes, nuts, seeds, and certain vegetables like broccoli and Brussels sprouts. Additionally, L-cysteine is available as a dietary supplement, often in the form of N-acetylcysteine (NAC), which is a more stable and bioavailable form.
Clinical Applications
N-Acetylcysteine (NAC)
N-acetylcysteine is widely used in clinical settings due to its mucolytic and antioxidant properties. It is commonly administered as a treatment for acetaminophen overdose, where it acts to replenish depleted glutathione levels and prevent liver damage. NAC is also used as a mucolytic agent in respiratory conditions such as chronic obstructive pulmonary disease (COPD) and cystic fibrosis.
Antioxidant Therapy
Given its role in glutathione synthesis, L-cysteine and its derivatives are being investigated for their potential in antioxidant therapy. This includes the management of conditions characterized by oxidative stress, such as neurodegenerative diseases, cardiovascular diseases, and diabetes.
Immune Function
L-cysteine has been shown to support immune function by enhancing the proliferation and activity of lymphocytes and other immune cells. This is partly due to its role in maintaining redox balance and protecting cells from oxidative damage.
Industrial Applications
L-cysteine is utilized in various industrial applications, including the food, pharmaceutical, and cosmetic industries. In the food industry, it is used as a dough conditioner in baking, as well as a flavor enhancer in meat products. In the pharmaceutical industry, L-cysteine is used in the production of certain drugs and as a stabilizing agent in formulations. In cosmetics, it is included in hair care products for its ability to strengthen hair and reduce damage.
Safety and Toxicity
L-cysteine is generally considered safe when consumed in amounts typically found in food. However, excessive intake, particularly in the form of supplements, can lead to adverse effects such as gastrointestinal distress, nausea, and vomiting. Long-term high-dose supplementation may also pose risks, including potential disruption of the body's redox balance and interference with mineral absorption.
Research and Future Directions
Ongoing research is exploring the potential therapeutic applications of L-cysteine and its derivatives in various medical conditions. This includes investigations into its role in cancer therapy, where it may enhance the efficacy of certain chemotherapeutic agents by modulating redox status. Additionally, research is examining the potential of L-cysteine in the treatment of psychiatric disorders, such as schizophrenia and bipolar disorder, where oxidative stress is implicated in disease pathology.
